Public Information
Public Information
Pharmacovigilance
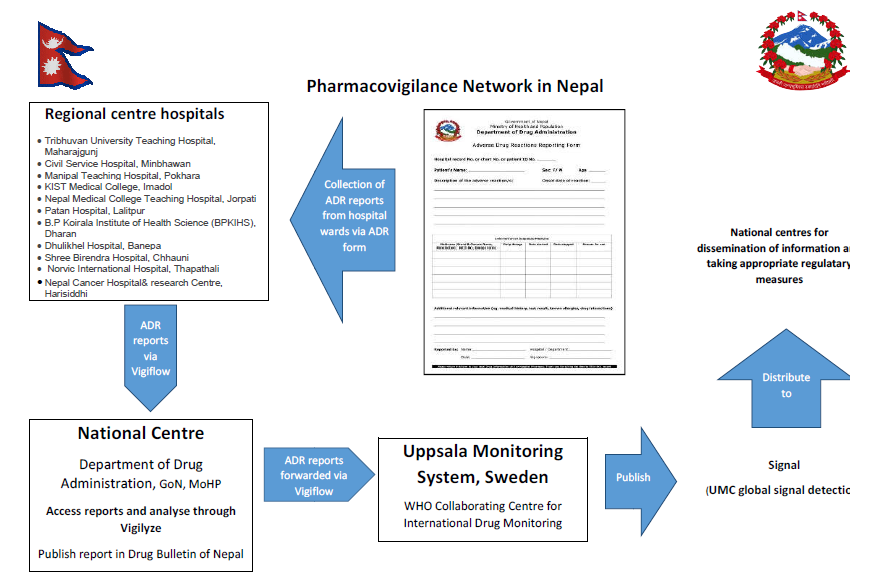
The Government of Nepal nominated Department of Drug Administration (DDA) in October 2004 as the focal point (National Pharmacovigilance centre) to liaison with WHO collaborating centre for International Drug Monitoring, Sweden and started collecting adverse drug reactions. Nepal became a WHO programme member in July 2006. In continuation to 15 regional pharmacovigilance centers, the department recently recognized 4 additional hospitals in Makwanpur, Chitwan, Bhaktapur, and Kathmandu to further strengthen the pharmacovigilance program.
At present, there are 17 regional pharmacovigilance centers in Nepal
- Tribhuvan University Teaching Hospital, Maharajgunj
- Civil Service Hospital, Minbhawan
- Manipal Teaching Hospital, Pokhara
- KIST Medical College, Imadol
- Nepal Medical College Teaching Hospital, Jorpati
- Patan Hospital, Lalitpur
- B.P Koirala Institute of Health Science (BPKIHS), Dharan
- Dhulikhel Hospital, Banepa
- Shree Birendra Hospital, Chhauni
- Norvic International Hospital, Thapathali
- Nepal Cancer Hospital and Research Center, Harisiddhi
- College of Medical Sciences - Teaching Hospital
- Nepal Mediciti, Kathmandu.
- Chitwan Medical College, Chitwan.
- National Tuberculosis Control Center, Bhaktapur.
- Hetauda Hospital, Makwanpur.
- Bharatpur Hospital, Chitwan.
- Bhaktapur Cancer Hospital, Bhaktapur.
- Kanti Children's Hospital, Maharajgunj.
These regional pharmacovigilance centers operate under DDA (DDA being the National centre for ADR monitoring). The regional centers reports ADRs to the National center (DDA) via ‘Vigiflow’ (an online program) which are then forwarded to the Uppsala Monitoring Center (UMC) by the National Centre. The National database contains about 1204 ADR reports so far.
Citizen Charter
Visit our citizen charter section for information on basic rules & regulation to acquire our service.
View Citizen CharterAny feedback?
Your feedback & suggestions are very important to us, Please send us your feedback!
Send Your Feedback